
Rare Disease CRO Services | Cromos Pharma
Introduction to Rare Disease CRO Support
A rare disease is defined differently across regulatory regions. Rare diseases affect an estimated 30 million people in the EU (roughly 1 in 17, or 4% of the population) and over 30 million in the U.S. (1 in 10 Americans). These definitions enable incentives such as market exclusivity, tax credits, and expedited regulatory pathways that support orphan drug development.
Cromos Pharma is an international full-service CRO supporting sponsors from first-in-human studies through post-marketing research. With operations across the US, Central & Eastern Europe, Central Asia, and Türkiye, we provide access to specialist centers, experienced investigators, and treatment-naïve patient populations for rare disease trials.
Our team combines global regulatory expertise with experience in complex studies, including advanced therapy medicinal products such as cell and gene therapies.
What this guide covers:
- Patient recruitment strategies for small populations
- Regulatory pathways and orphan drug designation support
- Innovative study design approaches for rare indications
- Advanced therapies including cell and gene therapies
- Real-world evidence and natural history studies
- Pediatric and family-centered trial operations
Alleviating the Burden of Rare Diseases
Behind every rare disease statistic is a patient or family facing a difficult journey. Many patients spend 5–7 years seeking a diagnosis, often consulting multiple specialists and facing misdiagnoses and significant emotional and financial strain. Most participants travel long distances to specialist centers while balancing care with work, school, and family life.
Common challenges include:
- Delayed diagnosis
- Limited specialist access
- Long travel distances
- Burdensome assessments
- Limited treatment options
Our patient-centric principles:
- Incorporate patient and caregiver feedback
- Design practical visit schedules
- Reduce unnecessary procedures
- Provide logistical support such as travel assistance
Hybrid trial approaches combining clinic and at-home assessments can further reduce burden and improve retention.
Rare Disease Clinical Trial Services
Cromos Pharma provides end-to-end rare disease CRO services, from feasibility assessments and protocol design to submission-ready clinical study reports. Rare disease trials require specialized operational models due to small, geographically dispersed patient populations and intensive follow-up requirements.
Cromos Pharma Rare Disease CRO Services:
- Feasibility and epidemiology assessments
- Protocol and endpoint strategy development
- Site selection and qualification
- Patient recruitment and retention programs
- Comprehensive trial execution
- Data management and biostatistics
- Pharmacovigilance and safety monitoring
- Medical writing and regulatory submissions (FDA, EMA, EU member states)
- Clinical trial supplies and logistics coordination
Our operating model is tailored for orphan indications, where site activation, patient support, and cross-border regulatory coordination are often more complex. We prioritize patient experience throughout the study to ensure participation remains manageable and supportive.
Patient-Centric Trial Design and Operations
A CRO should design rare disease studies to minimize visit burden and travel. This includes consolidating clinic visits, enabling local laboratory and imaging services where possible, and incorporating hybrid or decentralized trial elements. Addressing the specific needs of rare disease patients is essential, as many face barriers to accessing clinical trials.
Patient and caregiver input shapes our approach from the earliest stages through interviews, questionnaires, and advisory engagement.
Operational solutions include:
- Home health visits for infusions or sample collection
- Travel and accommodation support
- Translated consent forms and patient materials
- Flexible visit windows
- Childcare and meal support during longer visits
In rare disease trials, patient retention depends on reducing burden and respecting patients’ lives outside the clinic.
Global Reach and Regional Strength in Rare Disease Trials
Cromos Pharma operates across a geographic footprint selected for rare disease research success, with offices and site networks in the US, Central & Eastern Europe, Central Asia, and Türkiye.
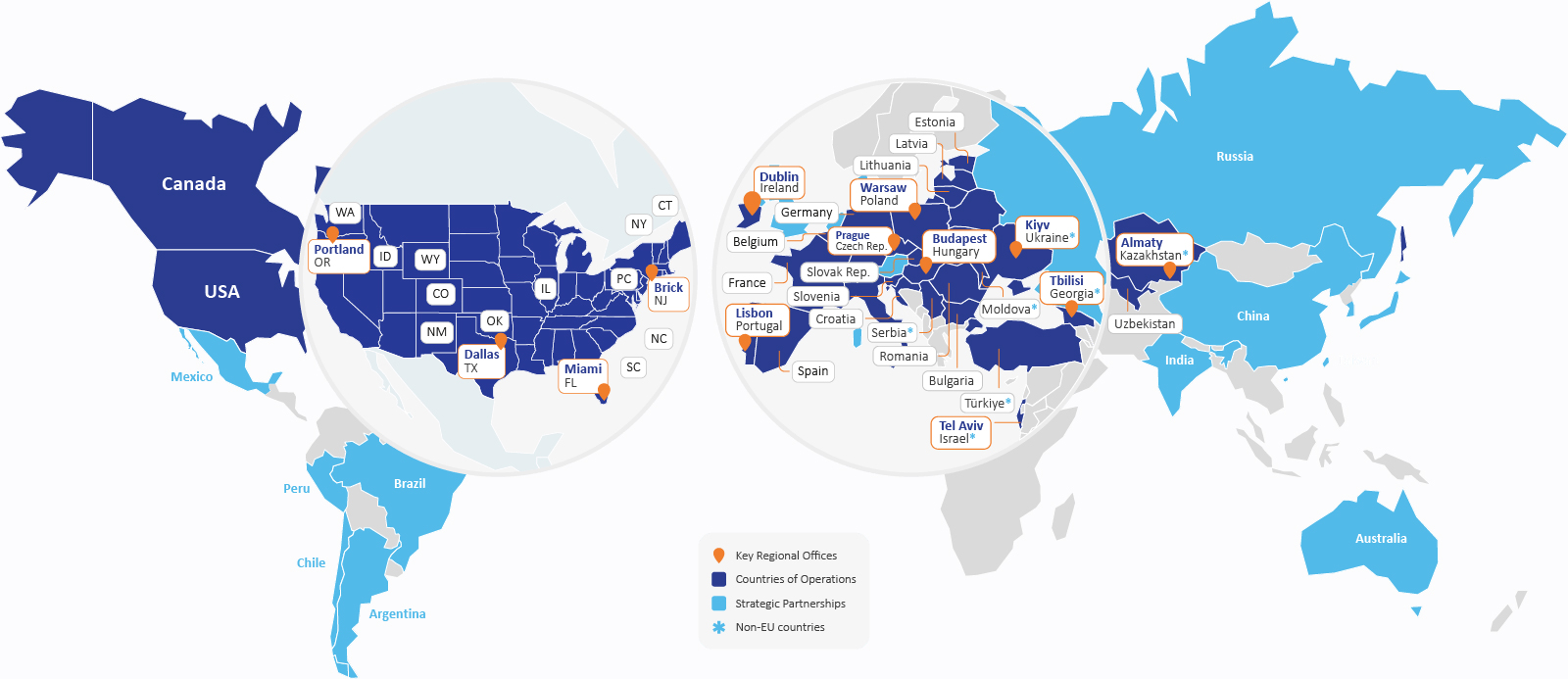
These regions offer important advantages. Centralized specialist centers often concentrate rare patients under the care of experienced investigators with strong patient relationships. Many of these sites also face less competition from other trials than major academic centers in Western Europe or the US, helping improve recruitment speed.
Benefits of our regional presence:
- Fast regulatory and site startup timelines
- High enrollment rates through established referral networks
- Strong patient retention via close investigator relationships
- Access to treatment-naïve populations
- Consistent quality across all countries
Expertise in Rare Disease Study Design and Methodology
Rare disease trials require innovative designs suited to small and heterogeneous populations. Traditional placebo-controlled studies may be impractical when patient numbers are extremely limited. In addition to innovative designs, rare disease research often involves managing complex clinical trials and navigating a complex regulatory landscape, especially with the rise of advanced therapy medicinal products (ATMPs) such as cell and gene therapies.
Common design approaches include:
- Adaptive trial designs
- Single-arm studies with external or historical controls
- Biomarker-based enrichment strategies
- Basket or umbrella trial concepts (innovative trial designs like basket and umbrella trials are being utilized to address complex considerations)
- Surrogate endpoints linked to clinical outcomes
Adaptive trial designs play a crucial role in optimizing efficacy and safety assessments in rare disease research.
Endpoints must remain scientifically robust while also being feasible for small patient populations. Selecting appropriate clinical endpoints and biomarkers is a significant challenge in designing rare disease clinical trials. Selecting the right endpoints to establish clinical benefit is one of the most challenging aspects of rare disease drug development.
Natural History and Real-World Evidence
Many rare diseases lack sufficient epidemiological and progression data, which creates challenges for study design and regulatory discussions. A rare diseases CRO should support natural history studies, registries, and real-world evidence (RWE) generation to address these gaps and inform clinical development.
Understanding disease progression, phenotypic variability, and baseline severity helps optimize endpoint selection and trial design.
Methodology | Purpose in Rare Disease Trials |
Retrospective chart reviews at expert centers | Understand historical disease progression |
Prospective observational cohorts | Track natural disease course over time |
Digital health tools (wearables, ePROs) | Capture real-world patient outcomes |
Registry analysis and data synthesis | Aggregate data across rare populations |
Precision Medicine and Biomarker-Driven Trials
Because about 80% of rare diseases have a genetic basis, genomics and molecular profiling are central to many development programs. Precision medicine approaches allow sponsors to target subpopulations where biology suggests a higher likelihood of benefit.
Challenges we help sponsors navigate include:
- Small genotype-defined subgroups requiring multi-country enrollment
- Assay standardization and validation across laboratories
- Genotype-phenotype variability affecting disease severity
- Regulatory requirements for companion diagnostic approval
Our goal is to ensure that biomarker-driven rare disease studies remain both scientifically rigorous and operationally feasible.
Patient Identification and Recruitment in Rare Disease Trials
Identifying and enrolling patients is one of the main challenges in rare disease trials, as populations are small, geographically dispersed, and often underdiagnosed. Standard recruitment models used for common diseases are rarely effective.
Cromos Pharma applies a targeted recruitment strategy based on feasibility assessments, collaboration with specialist centers, engagement with key opinion leaders, and outreach through patient organizations where permitted.
Our approach includes:
- Multi-country feasibility assessments
- Engagement with KOLs and principal investigators
- Referral networks to identify eligible patients
- Collaboration with registries and patient organizations
Decentralized and Hybrid Rare Disease Trials
A rare disease CRO may use decentralized and hybrid trial elements to make participation more feasible for patients who live far from specialist centers. These approaches reduce geographic barriers while maintaining data quality and regulatory compliance.
Examples of decentralized components include:
- Home nursing for infusions, injections, or sample collection
- Telemedicine visits for interim assessments
- Local laboratory testing coordinated with central labs
- Remote monitoring using wearables or home-based sensors
- ePRO platforms for symptom and quality-of-life reporting
When implementing remote components, data integrity and regulatory acceptability are carefully assessed. These approaches can reduce travel burden, improve patient retention, and expand access for rural or underserved populations.
Regulatory Strategy and Orphan Drug Development Pathways
Bringing a new therapy to patients is as much a regulatory journey as it is a scientific one. Every submission, meeting, and document must align perfectly with evolving international standards — from FDA INDs and NDAs to EMA CTIS, IMPDs, and the EU CTR framework.
At Cromos Pharma, we view regulatory affairs not as an administrative necessity but as a strategic driver that helps sponsors move from preclinical proof-of-concept to market authorization efficiently and compliantly. Our regulatory experts combine global insight with operational agility to help sponsors navigate multi-jurisdictional submissions, manage timelines, and achieve faster approvals.
Key regulatory services:
- Orphan drug designation preparation (FDA, EMA)
- IND/CTA submissions across multiple jurisdictions
- Scientific advice and protocol assistance preparation
- Expedited pathway applications (Fast Track, Breakthrough Therapy, Priority Review, PRIME)
Pediatric and Family-Centered Rare Disease Trials
Around 80% of rare diseases are genetic, and 50–75% affect children. Hence, pediatric populations are central to rare disease research. Pediatric trials are essential but complex due to small patient populations, severe conditions, and important ethical considerations. Engaging children and their families is critical to successful studies.
Key elements of our pediatric approach:
• Age-appropriate assent and consent materials
• Child-friendly visit environments with trained pediatric staff
• Flexible scheduling around school and family needs
• Psychological support during difficult procedures
• Careful management of blood draw volumes
• Age-appropriate and play-based outcome measures
We work closely with pediatric specialists and caregivers to ensure procedures and endpoints are feasible and appropriate for young patients.
Why Choose Cromos Pharma as Your Rare Disease CRO Partner
Developing therapies for rare and genetic diseases requires a CRO with specialized expertise and a flexible approach to complex clinical programs. Cromos Pharma supports sponsors with patient-centric trial design, access to recruitment-rich regions, full-service clinical development capabilities, and experience with innovative study designs tailored to small and geographically dispersed patient populations.Many sponsors choose a full service CRO to ensure faster timelines and better coordination across trial phases.
With our experience in global clinical research operations, integrated project management, data management, safety oversight, and strong site networks, Cromos Pharma helps sponsors navigate feasibility, regulatory strategy, and efficient trial execution in rare and orphan disease programs.






























