
Clinical Trials in Hungary – Country Profile 2022
Hungary boasts several key advantages for Sponsors considering the country as a location for clinical research. Its membership of the EU, high standards in education (in particular clinical and medical training), recent national programs aimed at developing a strong medical research sector, and general economic initiatives aimed attracting international investment all mark Hungary out as an attractive location for international Sponsors. Hungary is placed 10th worldwide in terms of the number of trials conducted in the country and 4th in Europe (based on population-proportional patient numbers). Each year approximately 20,000 patients participate in clinical trials in Hungary. Cromos Pharma established its office in Budapest, Hungary in 2016.
TOP 5 REASONS TO LOCATE YOUR CLINICAL TRIAL IN HUNGARY
- Hungary is an EU member state which has a centralized health system that assists rapid patient recruitment.
- It has a clear regulatory process and authorities that have been working to improve efficiencies, resulting in quick approval periods and expedited timelines.
- The country has a large pool of highly skilled and motivated medical professionals.
- Hungary offers excellent recruitment potential across a wide range of therapeutic areas especially for multi-country trials.
- Hungary has an excellent track record of producing high quality data as confirmed by positive inspection records from the FDA and EMA.
HEALTH OF THE NATION
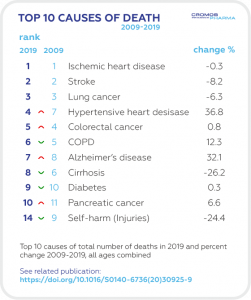 The average life expectancy at birth for males is 72 and for females is 79 years. This is significantly lower than the EU average life expectancy of 80.9 years (83.5 years for females and 78.3 for males). The two leading causes of death are malignant neoplasms (Hungary takes 1st place for the incidence of colorectal and lung cancer in the EU ranking list) and ischemic heart disease.
The average life expectancy at birth for males is 72 and for females is 79 years. This is significantly lower than the EU average life expectancy of 80.9 years (83.5 years for females and 78.3 for males). The two leading causes of death are malignant neoplasms (Hungary takes 1st place for the incidence of colorectal and lung cancer in the EU ranking list) and ischemic heart disease.
HUNGARY AT A GLANCE
- Hungary is a landlocked country located in Central Europe.
- It became a member state of the European Union in 2004.
- Population 9.75 million (2020).
- GDP $164 billion.
- Life Expectancy at Birth – 76 years.
- Largest city and capital is Budapest with a population of 1.75 million.
- Pharmaceutical sector is one of the main pillars of the Hungarian economy accounting for 7.5% of national GDP.
Source: World Bank
ADVANTAGES FOR SPONSORS
Significant naive patient population in a wide range of clinical indications
Hungary has a notable naive patient population in a wide range of therapeutic areas. Its patient
population is well disposed to taking part in clinical trials in order to get access to novel and advanced
treatments which may not be available to them through the national health system most notably in the
area of oncology.
Competitive cost of research
The cost of research in Hungary is very competitive when compared to neighboring western EU countries. In addition, investigator grants, supplementary vendor, and service costs are also highly competitive when compared worldwide.
Highly skilled clinical workforce producing high quality data
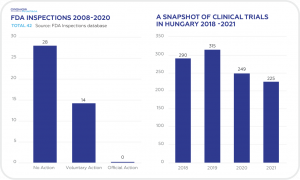
Hungary’s experience in clinical trials has led to the development of a highly skilled clinical workforce which has gained a reputation for producing high quality data. These high standards are demonstrated by the results of international inspections. For example, between 2008-2020 the US Food and Drug Administration (FDA) carried out a total of 42 inspections. The results of which were 28 No Action Indicated, 14 Voluntary Action Indicated and no Official Action Indicated.
Centralized health system and excellent infrastructure
Hungary has a centralized health care system, with four medical universities (Budapest, Debrecen, Szeged, Pécs) and over 160 governmental hospitals (approx. 100 active treatment centers and several
specialized/ rehabilitation institutions). Out of these, 18 specialise in conducting Phase I studies
(four of them are Phase I oncology centers). There are 66 private health care providers and the majority are engaged in clinical trials, most of them utilizing site management organizations (SMO). Several SMO networks are present in Hungary that are specialized in conducting clinical studies. Hungary has a tax-funded universal health care system organized through the National Health Insurance Fund . There is also a wide range of private health insurance schemes available to citizens. The centralized nature of Hungary’s health care system assists rapid patient recruitment.
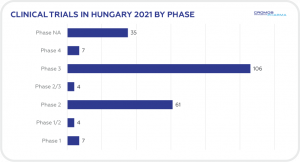
CLINICAL TRIALS INITIATED IN HUNGARY 2021
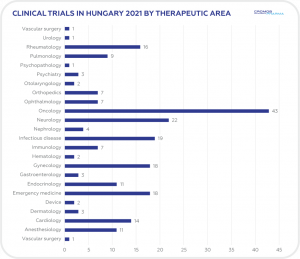 A total of 225 clinical trials were initiated in Hungary in 2021 according to data obtained from clinicaltrials.gov. This compares to 249 in 2020. The majority (106) were Phase 3 and oncology was the leading therapeutic area (43), followed by neurology (22), and infectious disease (19). International
A total of 225 clinical trials were initiated in Hungary in 2021 according to data obtained from clinicaltrials.gov. This compares to 249 in 2020. The majority (106) were Phase 3 and oncology was the leading therapeutic area (43), followed by neurology (22), and infectious disease (19). International
Sponsors represented 88% of trials initiated in 2021.

REGULATORY SYSTEM
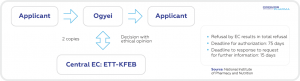
As a European Union member state since 2004, Hungarian laws and regulations are EU harmonized (e.g. EU CTD, GDPR). The relevant authorities with regard to the regulation of clinical trials are the National Institute of Pharmacy and Nutrition (NIPN) [Országos Gyógyszerészeti és Élelmezés-egészségügyi Intézet (OGYÉI)] working with the central ethics committee, and the Medical Research Council (MRC) [Egészségügyi Tudományos Tanács (ETT)]. The authorization process is well regulated including specialized regulations for Phase I, GMO, medical device and non-interventional studies. For instance, when a clinical trial is conducted with a GMO IMP, the Committee of Human Reproduction of MRC will also be involved in the initial authorization process.
OUTLINE OF CLINICAL TRIAL SUBMISSION PROCESS
During the clinical trial authorization process the NIPN/OGYÉI asks for the Specialized Authority
Position Statement from the Medical Research Council in parallel with its own review of the package
and will only authorize the trial where the central ethics committee provides a positive opinion
(i.e. a supportive Specialized Authority Position Statement).
The MRC has the following ethical committees:
- The Ethics Committee for Clinical Pharmacology
- The Committee of Human Reproduction
- The Scientific and Research Committee
- The System of Regional Committee
The local, or regional ethics committees do not authorize studies, they are responsible for local oversight of the trials and need information notification of the study start-up, close-out and about the local SAEs.
The NIPN/ OGYÉI and the MRC have a reputation for being responsive during the clinical trial submissions and authorization processes as compared to other neighboring countries and several other regions of the world. They makes for a highly collaborative and supportive regulatory environment.
Currently there are two ways for the initial authorization of a trial via:
- National Authorization
- Voluntary Harmonization Process (VHP)
In general, using the national process is more favorable in the case of single country trials. The VHP can be used in multi- country trials involving at least two EU countries. VHP is a procedure which makes it possible to obtain a coordinated assessment of an application for a clinical trial that is to take place in several European countries 25. It harmonizes the approval timelines of key documents, like protocol, IB, IMPD among the participating countries. Hungary has actively taken part (since 2009) in the VHP process. The Hungarian competent authority is often involved as the reference country, therefore it has significant experience with this authorization procedure.
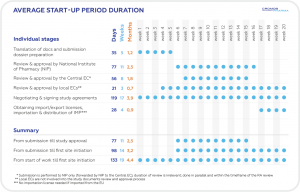
AUTHORIZATION TIMELINES BASED ON CROMOS PHARMA-MANAGED TRIALS (2018-2021)
 After the receipt of core documents in English (incl. preparation of the submission package with customization and preparation of the Clinical Trial Application (CTA or Annex I) form, translations and customization of ICF, PIS, patient card, label, synopsis, patient diaries, and questionnaires/PROs).
After the receipt of core documents in English (incl. preparation of the submission package with customization and preparation of the Clinical Trial Application (CTA or Annex I) form, translations and customization of ICF, PIS, patient card, label, synopsis, patient diaries, and questionnaires/PROs).

EU CLINICAL TRIAL REGULATION
The EU Clinical Trial Regulation (Regulation (EU) No 536/2014), which came into effect at the start of 2022 implements a VHP-like centralized authorization process for all EU member states. Hungary is in a good position to adapt to this new system having gained significant experience in the VHP process in recent years.
CASE STUDY
In 2018, Cromos Pharma was contracted to rescue an ongoing Phase I/II multi-country clinical trial in the field of neurology with recruitment di2culties. Hungary was selected to support the recruitment. Hungarian sites demonstrated an exceptional contribution by providing more than 50% of enrolled subjects.
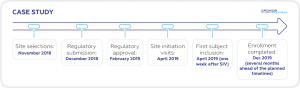
Following a quick start-up period (~4.5 months from start of submission preparation to first
site initiation visit), the study accrual increased significantly meeting the enrollment goal
ahead of the client’s expected timelines.
ABOUT CROMOS PHARMA
Cromos Pharma provides tailored and effective clinical trial services to support the development of drugs that transform healthcare. A US-based international CRO, with over 18 years’ experience offering fully integrated services and delivering all aspects of clinical trials in all clinical phases across a wide range of therapeutic areas. Cromos Pharma delivers rapid recruitment and excellent patient retention as well as expert study design and management. Cromos Pharma has strong regional experience in Central and Eastern Europe. Its US HQ is in Portland, Oregon and its European HQ is situated in Dublin, Ireland.
CROMOS PHARMA IN HUNGARY
Cromos Pharma has an experienced team on the ground in Budapest who effectively manage regulatory and contracting processes to ensure that studies can get up and running in the quickest time possible. We recruit highly educated and experienced personnel to guarantee each trial managed by our team in Hungary produces the exceptional quality data and results.
Read our PDF here.
For more information about running your clinical trial in Hungary please contact Zsolt Kocsis, MD, MBA Head of Region, Central Europe zsolt.kocsis@cromospharma.com.
Cromos Pharma proudly supports the Right2hope nonprofit platform, empowering patients to explore treatment options beyond traditional clinical trials. Through an intuitive, AI-supported matching algorithm, Right2hope connects patients with the most relevant expanded access, compassionate use, and right-to-try programs.






























