
FDA Signals Shift in Regulatory Approach to Autoimmune CAR-T
Recent signals from the US FDA suggest regulators are preparing a more tailored approach for CAR-T therapies in autoimmune diseases. As the field rapidly evolves, the agency has indicated it intends to carefully guide development while paying close attention to long-term safety and durability of response.
Regulators have also suggested that development pathways may need to be evaluated on a case-by-case basis depending on disease severity and patient population, while highlighting potential risks such as unpredictable long-term toxicity and possible fertility effects that will require careful monitoring.
The timing of this shift is not accidental. Over the past two years, the clinical evidence for autoimmune CAR-T has moved from striking case reports to early trials that are beginning to resemble pivotal-intent programs.
What once looked like a scientific curiosity is now becoming a serious development pathway.
From Astonishing Case Reports to Structured Trials
The modern autoimmune CAR-T story began with small but remarkable clinical observations.
A 2024 case series published in The New England Journal of Medicine described 15 patients with severe autoimmune diseases including systemic lupus erythematosus, inflammatory myopathies, and systemic sclerosis who received a single CD19-directed CAR-T infusion. Many patients achieved remission and were able to discontinue immunosuppressive therapy.
For diseases typically managed with lifelong immunosuppression, the results were striking.
Subsequent early-phase studies began expanding the evidence base. A multicenter phase 1 study of BCMA-CD19 compound CAR-T therapy in systemic lupus erythematosus with lupus nephritis reported elimination of circulating autoantibodies and major reductions in disease activity scores, with medication-free remission observed in most treated patients and follow-up extending up to 46 months.
Since then the field has progressed into more structured studies. The CASTLE basket trial enrolled patients with severe treatment-refractory autoimmune diseases and reported sustained immunosuppression-free disease control with relatively manageable safety signals.
While these studies remain small, the biological signal is increasingly difficult to ignore. For the first time, CAR-T is being seriously discussed as a therapy that could reset immune function rather than simply suppress autoimmune disease.
At the same time, the broader industry is rapidly mobilizing around the concept. More than a dozen biotechnology companies are now developing CAR-T programs targeting autoimmune conditions including lupus, multiple sclerosis, myositis, and rare neurologic disorders.
One of the most advanced programs may soon test the regulatory pathway. Kyverna Therapeutics has reported positive pivotal trial results for its CD19-directed CAR-T therapy mivocabtagene autoleucel for stiff person syndrome and is preparing a potential FDA submission in 2026.
Why Regulators Are Moving Carefully
Despite the excitement, regulators are approaching autoimmune CAR-T development cautiously. The reason lies in a fundamental difference between oncology and autoimmune disease.
Most CAR-T therapies in oncology are used in patients with life-threatening cancers who have exhausted other treatment options. In autoimmune diseases the population is very different. Many patients are younger, may be of reproductive age, and may live with their condition for decades. This changes the risk-benefit equation.
Because CAR-T products are classified as gene therapies in the US, regulators already require long-term follow-up in treated patients. Current guidance recommends monitoring for as long as 15 years after treatment when integrated genetic material is involved.
Recent regulatory actions reinforce this cautious stance. In 2024 the FDA required updates to the boxed warning across CD19- and BCMA-directed CAR-T therapies after reports of secondary T-cell malignancies and recommended lifelong monitoring for secondary cancers.
At the same time, regulators are signaling flexibility where safety management is established. In 2025 the FDA eliminated the REMS requirement for several approved CAR-T therapies while maintaining long-term safety monitoring expectations.
Together these signals suggest that autoimmune CAR-T development will likely move forward with regulatory flexibility paired with strong long-term safety guardrails.
Regulatory Signals for Autoimmune CAR-T (2024–2026)
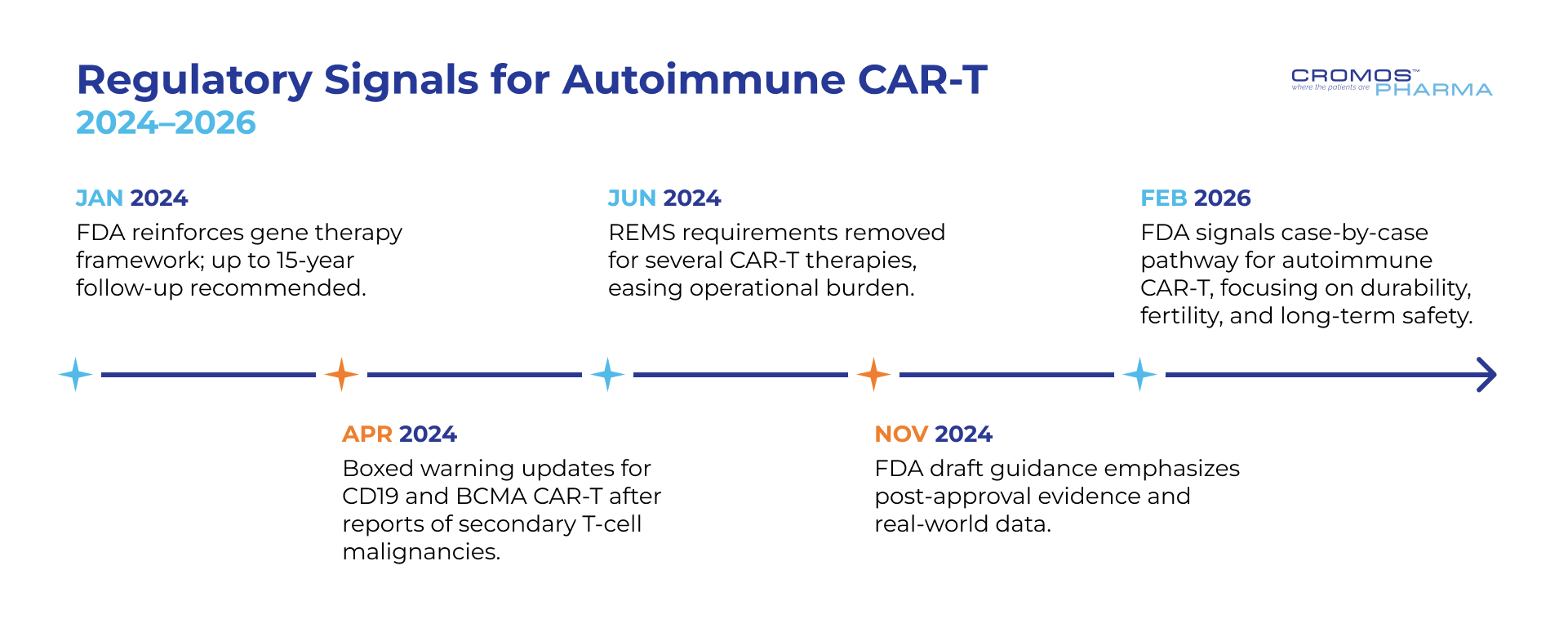
- Jan 2024 – FDA reinforces gene therapy framework; up to 15-year follow-up recommended.
- Apr 2024 – Boxed warning updates for CD19 and BCMA CAR-T after reports of secondary T-cell malignancies.
- Jun 2025 – REMS requirements removed for several CAR-T therapies, easing operational burden.
- Nov 2025 – FDA draft guidance emphasizes post-approval evidence and real-world data.
- Feb 2026 – FDA signals case-by-case pathway for autoimmune CAR-T, focusing on durability, fertility, and long-term safety.
The Next Challenge: Proving Durability
As development moves forward, the central question for autoimmune CAR-T will be durability.
Many of the most encouraging studies currently report follow-up periods of roughly one to two years. These results are promising but still leave important questions unanswered.
- Will remission persist over many years?
- Will relapse eventually occur as B-cell populations recover?
- And can the safety profile remain acceptable in patients expected to live for decades after treatment?
Another key question is whether CAR-T truly changes the course of disease or simply produces temporary suppression of autoimmune activity.
To answer this, future trials will likely rely on validated disease-specific endpoints such as DORIS remission criteria in lupus, ACR-EULAR outcomes in inflammatory myopathies, and functional measures in neurologic diseases, combined with organ-level outcomes.
In practice, many programs are expected to combine short-term efficacy endpoints with extended follow-up and structured long-term monitoring, reflecting the gene therapy–like safety expectations associated with CAR-T treatments. Only with this type of evidence will regulators, clinicians, and payers be confident that one-time cellular therapies can deliver durable benefit in chronic autoimmune diseases.
The Operational Reality
Beyond the scientific and regulatory questions, autoimmune CAR-T trials present significant operational challenges.
Key operational friction points include:
- Patient selection and referral pathways. Recruitment often depends on specialist referral networks and centers of excellence rather than large site lists, especially in severe refractory autoimmune disease.
- Manufacturing and logistics. Coordinating leukapheresis, cell processing, chain-of-identity tracking, and infusion timelines requires tightly integrated operational systems.
- Disease management during manufacturing. Patients may experience disease flares between leukapheresis and infusion if background therapies are paused, making bridging strategies and careful clinical monitoring essential.
- Long-term follow-up infrastructure. Because CAR-T therapies are regulated similarly to gene therapies, trials must support extended safety monitoring and data collection for many years.
For sponsors, autoimmune CAR-T development is therefore not only a scientific challenge but a clinical operations stress test requiring specialized infrastructure, experienced sites, and disciplined trial execution.
The Bottom Line
Autoimmune CAR-T is entering a pivotal stage of development. The scientific signal is strong enough to attract serious investment and regulatory attention, but the path forward will require careful trial design, long-term safety monitoring, and disciplined clinical execution.
Regulators appear open to innovation in this space. At the same time, they are making it clear that durable benefit and lifelong risk management will be central to the approval pathway.
The coming years will determine whether CAR-T becomes not only a breakthrough in oncology but also a transformative platform for autoimmune disease treatment.






























