
Clinical Trials in the Czech Republic: A Prominent Hub for Scientific Discovery and Cost Optimization
The Czech Republic has emerged as an attractive destination for clinical trials, offering a strong infrastructure, supportive regulatory environment, and ethical standards. This article provides a detailed exploration of the country’s scientific and logistical advantages, highlighting its cost- and time-saving potential. Additionally, the article presents a country overview, demographics, healthcare system, and the reasons why the Czech Republic is an ideal choice for conducting clinical research.
The Czech Republic’s geographical location in Central and Eastern Europe makes it a convenient and accessible destination for both local and international research. Its well-connected transportation infrastructure further facilitates the smooth conduct of clinical trials.
The country boasts a well-developed network of research institutions, hospitals, and private clinics. This infrastructure provides an ideal environment for clinical trials to take place, with access to state-of-the-art facilities, skilled medical professionals, and a concentrated pool of potential participants.
The country’s regulatory bodies prioritize patient safety and privacy while also promoting scientific progress. Their efficient and transparent approval processes facilitate the initiation and execution of clinical trials, encouraging researchers to choose the Czech Republic as a favorable destination for their studies.
The Czech Republic places a strong emphasis on ethical considerations in clinical research. Regulatory bodies ensure that trials are conducted with utmost integrity and adhere to strict ethical guidelines. This commitment to ethical standards helps maintain the trust of both patients and researchers.
In addition to its scientific and logistical advantages, Czech Republic offers significant cost savings compared to Western European countries and the United States. The lower costs associated with conducting clinical trials in Czech Republic stem from favorable Investigator Fees, competitive operational expenses, and lower patient recruitment expenses. These factors make the country an attractive option for biotech companies seeking to optimize their research and development budgets without compromising on quality.
Country Overview
The Czech Republic, also known as Czechia, is a landlocked country located in Central Europe. It is bordered by Germany to the west, Austria to the south, Slovakia to the east, and Poland to the north.
The capital of the Czech Republic is Prague, which is also the largest city in the country. Prague is renowned for its stunning architecture, historical landmarks, and vibrant cultural scene.
The Czech Republic has a highly developed and export-oriented economy. It is one of the most industrialized countries in Central Europe, with a strong focus on manufacturing, automotive, electronics, and machinery sectors. The country’s strategic location and skilled workforce attract robust foreign investments.
Czech is the official language of the country. English and German are also widely spoken, especially in urban areas and among the younger population. The official currency of the Czech Republic is the Czech Koruna (CZK).
The country is a member of the European Union, the United Nations, and the World Trade Organization.
Demographics
The Czech Republic has a relatively small population compared to other European countries. As of 2023, the estimated population is around 10.5 million people.
The Czech Republic exhibits a comparatively mature population, with a median age of approximately 43 years. The age structure shows a fair distribution, featuring a notable percentage of individuals in their middle and senior age groups. The 15-64 years age group constitutes the working-age population and accounts for around 65.2% of the total population.
The majority of the population in the Czech Republic is ethnically Czech, accounting for about 64% of the total population. There are also significant minorities of Slovaks, Moravians, and other ethnic groups.
The Czech Republic is highly urbanized, with a significant percentage of the population living in cities and metropolitan areas.
Healthcare System
The Czech Republic operates a universal healthcare system, which means that all citizens and legal residents have access to essential healthcare services. The system is funded through mandatory health insurance contributions, which are deducted from employees’ salaries and contributions from self-employed individuals.
The country has a network of hospitals, clinics, and healthcare centers, both in urban and rural areas. Major cities, including Prague, have well-equipped medical facilities and specialized treatment centers, while smaller towns and rural regions also offer healthcare services, although they may be more limited.
The healthcare system in the Czech Republic generally offers a high standard of care. The country has well-trained medical professionals, and many doctors and healthcare staff speak English and other languages.
Reasons to Conduct Clinical Trials in the Czech Republic
- Reliable clinical research infrastructure
- High density of clinical sites
- High educational standards, particularly in clinical medicine
- Well-organized centralized healthcare system
- Supportive regulatory framework
- Large pool of skilled clinical professionals trained in GCP delivering high quality data
- Considerable treatment-naïve patient populations in a diverse range of therapeutic indications
Snapshot of Czech Republic’s Clinical Trials
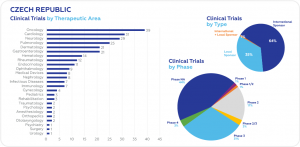
A total of 265 clinical trials were initiated between 1 July 2022 and 1 July 2023. According to the data from clinicaltrials.gov, the largest number of clinical trials were conducted in oncology (39), followed by cardiology (31), neurology (29), and pulmonology (25). International sponsors conduct 64% of all ongoing clinical trials.
Czech Republic: Quick Facts
Regulatory approval process
The application must be uploaded onto the Clinical Trials Information System (CTIS). Parts I and II of the documents are assessed in parallel within 45 calendar days (60 calendar days in total for review and approval).
Agreements with sites and investigators
Usually, 2 agreements should be signed – with an institution and with a PI. The draft contract must be submitted and if not available, statements regarding study funding and financial operations in the study must be submitted. Having a country-specific Clinical Trial Agreement template is helpful in the negotiation process.
Some institutions also allow for a three-party contract – between the sponsor/CRO, the institution and the investigator. In this case, a separate contract with the investigator is not necessary.
Since investigational drugs must be supplied to the pharmacy of choice under Czech law, a separate contract with the pharmacy is required. If the pharmacy is part of an institution, the contract with the pharmacy may be part of the contract with the institution.
If the institution does not have the ability to perform certain procedures in-house (e.g., MRI, CT, etc.), a separate contract may also need to be entered into with the supplier of those services.
EC review and approval
CEC is the only ethics body involved in the review process (Part II Ethical Review). Submission should be done via Clinical Trials Information System (CTIS).
Favorite trial sites’ locations
Main medical centers are located in major cities such as Prague, Ostrava, Brno, Plzen, Hradec Kralove, Olomouc.
Legal entity
For non-EU Sponsors, an EU legal representative is required. CTIS application form contains sections that should be filled out in the Czech language. Submission to CEC needs to be done in either Czech or in both English and Czech. The assistance of local staff is therefore recommended.
QP Declaration / GMP certificate
Declaration from the QP stating that the manufacturing site operates in compliance with the EU GMP is required for submission.
Documents requiring special attention
Informed Consent Form (ICF) should be completed in the local language and should contain country-specific information. For details, see guideline KLH-22 issued by the State Institute for Drug Control.
Official language
Essential documentation should be submitted in English. Patient-related documents, labels, protocol synopsis must be translated into the Czech language.
Patient insurance
Global insurance agreement with the local legal entity in the country is required. A further requirement is that a local contact who speaks the Czech language should be available to assist the patient with any questions.
Useful tips
Initial submissions made before 31 January 2023 and submitted under the old legislation (Clinical Trial Directive 2001/20/EC) can continue, based on the assumption that the trials will be completed by January 30, 2025. Processes will remain unchanged, and sponsors will therefore be able to submit substantial amendments and end-of-trial notifications as required under the Clinical Trial Directive.
Regulatory Environment for Clinical Trials in the Czech Republic
On 31 January 2022, the EU Clinical Trials Regulation No 536/2014 (CTR) became applicable in all EU/ European Economic Area (EEA) Member States, thereby replacing the EU Clinical Trials Directive (2001/20/EC) (CTD).
Effective January 31, 2023, all new CTAs must be submitted under the EU Clinical Trials Regulation via CTIS.
To perform clinical trials in the Czech Republic, the CTR requires that all CTIS User Administrator/User submit a harmonized format of the application dossier consisting of Part I (Scientific Review documents) and Part II (Ethical Review documents).
The Clinical Trials Regulation No 536/2014, Annex I, requires that a comprehensive list of documents is submitted. The CTIS User Administrator/User can submit Part I and Part II either simultaneously or sequentially.
The General timeline from study submission until initial approval is 60 days (validation – 10 days; assessment – 45 days; decision – 5 days). Parts I and II can be assessed in parallel within 45 calendar days and up to 76 calendar days if there are any requests for information.
Conclusion
The Czech Republic has emerged as an attractive choice for conducting clinical trials in the biotech industry. From its supportive regulatory environment and engaged patient population to state-of-the-art infrastructure and cost-effectiveness, the country offers biotech professionals vast opportunities for transformative research.
As the Czech Republic continues to evolve its clinical trial landscape, it is poised to make significant contributions to the global biotech industry’s progress, ultimately improving patient outcomes worldwide.
Cromos Pharma – your partner in International Clinical Trials
Cromos Pharma has an experienced local team that effectively manages regulatory and contracting processes to ensure that studies can be initiated in the shortest period possible. We recruit highly educated and experienced staff that assures that each trial managed by our team in the Czech Republic produces exceptional data quality and reliable results.
Cromos Pharma combines global expertise with in-depth local experience and knowledge that translates into exceptional patient recruitment. Our team has met or reduced enrollment timelines in 95% of the conducted trials.
If you are considering conducting clinical trials in the Czech Republic or in any of the other countries where Cromos Pharma is present, our team will be happy to answer your questions.
Cromos Pharma proudly supports the Right2hope nonprofit platform, empowering patients to explore treatment options beyond traditional clinical trials. Through an intuitive, AI-supported matching algorithm, Right2hope connects patients with the most relevant expanded access, compassionate use, and right-to-try programs.






























